understanding
|
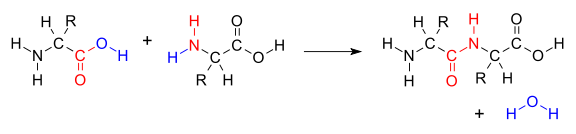
Carbohydrates and lipidsCondensation Reactions
a large molecule. Also water is formed in the reaction
|
applications
- Structure and function of cellulose and starch in plants and glycogen in humans
- Scientific evidence for health risks of trans-fats and saturated fats
- Lipids are more suitable for long-term energy storage in humans than carbohydrates
- Evaluation of evidence and the methods used to obtain evidence for health claims made about lipids
nature of science
- Evaluating claims: health claims made about lipids need to be assessed
Skills
- Use of molecular visualization software to compare cellulose, starch, and glycogen
- Determination of body mass index by calculation or use of a nomogram
A Polypeptide are condensation reactions link amino acids to either end of the dipeptide, eventuslly
forming a chain of amino acids
A Polysaccharide are two monosaccharides that can be linked to form a disaccharide and more
monosaccharides can be linked to a disaccharide to form a large molecule
A Triglyceride are a maximum of three fatty acids that can be linked to each glycerol
Hydrolysis Reactions
POLYPEPTIDES + WATER --------> DIPEPTIDES OR AMINO ACIDS
POLYSACCHARIDES + WATER --------> DISACCHARIDES OR MONOSACCHARIDES
GLYCERIDES + WATER --------> FATTY ACIDS + GLYCEROL
Examples of Carbohydrates
Examples Example of use in animals Example of use in plants
Monosaccharides Glucose Glucose is carried by the blood Frucotse is used to make fruits
Galactose to transport energy to cells sweet-tasting, attracting animals
Fructose throughout the body to disperse seed in the fruit
Disaccharides Maltose Lactose is the sugar in milk, Sucrose is carried by phloem to
Lactose that provides energy to young transport energy to cells throu
Sucrose mammals until they are weaned the plant
Polysaccharides Starch Glycogen is used as ashort-term Cellulose is ised to make strong fibres
Glycogen energy store in liver and in muscles that are used to construct the plant cell
Cellulose wall
Functions of Lipids
Carbohydrates and Lipids in energy storage
forming a chain of amino acids
A Polysaccharide are two monosaccharides that can be linked to form a disaccharide and more
monosaccharides can be linked to a disaccharide to form a large molecule
A Triglyceride are a maximum of three fatty acids that can be linked to each glycerol
Hydrolysis Reactions
- Large molecules such as polypeptides, polysaccharides and triglycerides can be
- Water molecules are used up in hydrolisis reactions
- Hydrolisis reactions are the reverse of condensation reactions
POLYPEPTIDES + WATER --------> DIPEPTIDES OR AMINO ACIDS
POLYSACCHARIDES + WATER --------> DISACCHARIDES OR MONOSACCHARIDES
GLYCERIDES + WATER --------> FATTY ACIDS + GLYCEROL
Examples of Carbohydrates
Examples Example of use in animals Example of use in plants
Monosaccharides Glucose Glucose is carried by the blood Frucotse is used to make fruits
Galactose to transport energy to cells sweet-tasting, attracting animals
Fructose throughout the body to disperse seed in the fruit
Disaccharides Maltose Lactose is the sugar in milk, Sucrose is carried by phloem to
Lactose that provides energy to young transport energy to cells throu
Sucrose mammals until they are weaned the plant
Polysaccharides Starch Glycogen is used as ashort-term Cellulose is ised to make strong fibres
Glycogen energy store in liver and in muscles that are used to construct the plant cell
Cellulose wall
Functions of Lipids
- ipids can be used for energy storage in the form of fat in humans and oil in plants.
- Lipids can be used as heat insulation as fat under the skin reduces heat loss.
- Lipids allow buoyancy as they are less dense than water and so animals can float in water
Carbohydrates and Lipids in energy storage
Cellulose Starch Glycogen
Link together beta glucose Link together alpha glucose Link together alpha glucose
(OH on top of the ring for carbon 1) (OH on bottom of the ring for carbon 1) Highly branched
Curved Straight Storage molecule
Moderately branched High tensile strength Animal
Storage molecule Cell Wall
Plant Plant
Link together beta glucose Link together alpha glucose Link together alpha glucose
(OH on top of the ring for carbon 1) (OH on bottom of the ring for carbon 1) Highly branched
Curved Straight Storage molecule
Moderately branched High tensile strength Animal
Storage molecule Cell Wall
Plant Plant
Body mass index
- Developed by Adolph Quetelet (1830s)
- By calculation or use of a nomogram
trans/cis isomers
•Unsaturated fats can be trans/cis isomers
•Cis: liquid at room temperature (oil)
•Trans: solid at room temperature (margarine)
•There are potential health risks for trans-unsaturated and saturated fats
•Cis: liquid at room temperature (oil)
•Trans: solid at room temperature (margarine)
•There are potential health risks for trans-unsaturated and saturated fats
Lipids
- Condensaton of three fatty acids and glycerol form a Tryglyceride .
Sources
Molecular Biology Notes
Study Guide Biology
Pearson Textbook
Molecular Biology Notes
Study Guide Biology
Pearson Textbook
TOK QUESTIONS
There are conflicting views as to the harms and benefits of fats in diets. How do we decide between competing views?
Of course, fats in diets have their harms and benefits. There are two main types of potentially harmful dietary fat — fat that is mostly saturated and fat that contains trans fat. The types of potentially helpful dietary fat are mostly unsaturated: Monounsaturated fat, Polyunsaturated fat, and Omega 3 fatty acids.
Of course, fats in diets have their harms and benefits. There are two main types of potentially harmful dietary fat — fat that is mostly saturated and fat that contains trans fat. The types of potentially helpful dietary fat are mostly unsaturated: Monounsaturated fat, Polyunsaturated fat, and Omega 3 fatty acids.